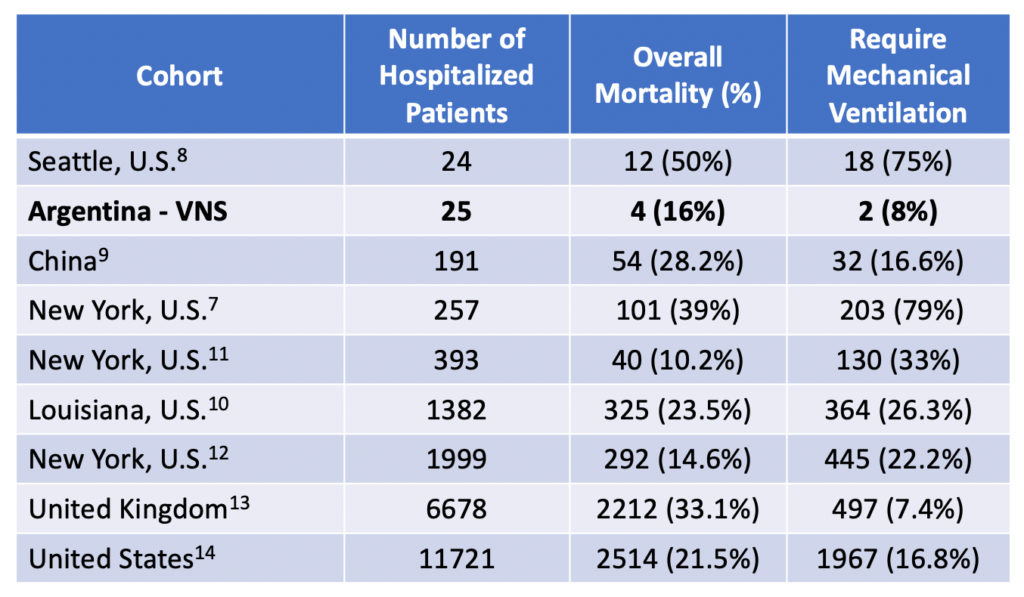
OVERLAND PARK, KANSAS, UNITED STATES, December 2, 2020 /EINPresswire.com/ — Nemechek Technologies is proud to announce publication of a peer-reviewed research article by its CEO, Patrick Nemechek D.O. Entitled, ‘Transcutaneous Vagus Nerve Stimulation is Associated with Lower Mechanical Ventilation and Mortality in COVID-19 Patients: An interim Safety Analysis’, the study reveals that transcutaneous vagus nerve stimulation (tVNS) is safe and potentially beneficial for hospitalized patients with COVID-19 pneumonia. Patients meeting clinical criteria received tVNS therapy for 5 minutes 2-4 times per day.
Patient outcomes included infrequent use of mechanical ventilation, and a high rate of survival. Interim analysis found the mortality rate and the frequency of mechanical ventilation to be less than almost all other large COVID-19 cohorts. The study, an ongoing single-arm, uncontrolled open-label, observational trial to assess (tVNS) in hospitalized SARS-CoV-2 infected subjects, is being conducted at Hospital Zonal Virgen Del Carmen in Zarate, Buenos Aires, Argentina.
“We are extremely encouraged by the reduced mortality in patients receiving tVNS therapy. The trial is moving forward to achieve its planned total enrollment, and we will publish further results once all 50 subjects have been treated.” – Patrick Nemechek D.O.
About Nemechek Technologies, LLC
Nemechek Technologies, LLC is a privately held manufacturer of bio-electronic medicine. Their first product, the Vitality Smartcable®, is a consumer wellness product.
With 20 years’ experience as an HIV physician and researcher, Dr. Nemechek was awarded U.S. Patent No. 10,335,396 for his method of lowering inflammation and reversing autonomic nervous system damage. The Nemechek Protocol®, which focuses on reducing chronic inflammation, includes the use of transcutaneous vagus nerve stimulation. Dr. Nemechek has treated more than 1,000 patients with vagus nerve stimulation therapy over the past ten years at his clinic near Phoenix, Ariz.
Mike Nemechek
Nemechek Technologies, LLC
+1 913-219-1239